- Product Name
- CasNo
- MF
- MW
- Content
- Appearance
- Packing
- Apply
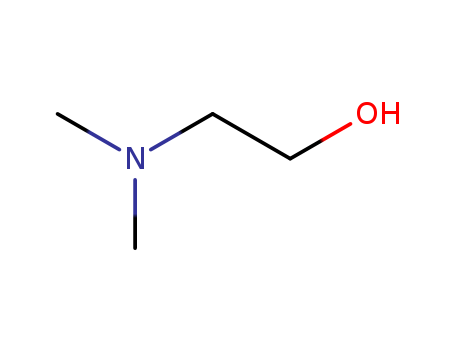
- N,N-Dimethylethanolamine?(DMEA)
- 108-01-0
- C4H11NO
- 89.1374
- clear to pale yellow liquid
Your Location:Home > Products > Catalysts and additives > N,N-Dimethylethanolamine?(DMEA)

|
Preparation |
The synthesis of 2-Dimethylaminoethanol by the ethylene oxide method is obtained by the ammonification of dimethylamine with ethylene oxide, which is distilled, refined and dehydrated. |
|
Production Methods |
Synthesis of dimethylaminoethanol can be accomplished from equimolar amounts of ethylene oxide and dimethylamine (HSDB 1988). |
|
Air & Water Reactions |
Flammable. Partially soluble in water and less dense than water. |
|
Reactivity Profile |
DIMETHYLAMINOETHANOL is an aminoalcohol. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. N,N-Dimethylethanolamine may react vigorously with oxidizing materials. |
|
Health Hazard |
Dimethylaminoethanol is classified as a mild skin irritant and a severe eye irritant (HSDB 1988). Doses as high as 1200 mg daily produce no serious side effects and a single dose of 2500 mg taken in a suicide attempt had no adverse effects (Gosselin et al 1976). Inhalation of the vapor or mist can cause irritation to the upper respiratory tract. Asthmatic symptoms have been reported. Extremely irritating; may cause permanent eye injury. Corrosive; will cause severe skin damage with burns and blistering. Ingestion may cause damage to the mucous membranes and gastrointestinal tract. No reports were found in the literature regarding carcinogenic or mutagenic potential. |
|
Flammability and Explosibility |
Flammable |
|
Safety Profile |
Moderately toxic by ingestion, inhalation, skin contact, intraperitoneal, and subcutaneous routes. A skin and severe eye irritant. Used medically as a central nervous system stimulant. Flammable liquid when exposed to heat or flame; can react vigorously with oxidzing materials. Ignites spontaneously in contact with cellulose nitrate of high surface area. To fight fire, use alcohol foam, foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx |
|
Metabolism |
When administered orally, dimethylaminoethanol acetamidobenzoate (the therapeutic salt formulation) has been shown to cross the blood-brain barrier (HSDB 1988). Two other studies have examined the pharmacokinetics of dimethylaminoethanol in rats (Dormand et al 1975) and healthy adults (Bismut et al 1986). It has been postulated that dimethylaminoethanol undergoes endogenous methylation (LaDu et al 1971). After intravenous treatment of mice with [14C]-labeled dimethylaminoethanol in the brain, dimethylaminoethanol yielded phosphoryldimethylaminoethanol and phosphatidyldimethylaminoethanol. Acid-soluble and lipid cholines derived from dimethylaminoethanol also were found in brain (Miyazaki et al 1976). While examining the pharmacokinetics of the maleate acid of [14C]-dimethylaminoethanol in rats, Dormand et al (1975) observed that dimethylaminoethanol was metabolized in the phospholipid cycle and produced metabolites such as phosphoryldimethylaminoethanolamine, and glycerophosphatidylcholine. In kainic-acid lesioned rats, dimethylaminoethanol was converted to a substance which cross-reacted in the radioenzymatic assay for acetylcholine (London et al 1978). Ansell and Spanner (1979) demonstrated that [14C]-dimethylaminoethanol rapidly disappeared from brain; after 0.5, 1, and 7 h, only 30, 27, and 16% of the administered radioactivity, respectively, remained in the brain after intracerebral injection. They also showed that brain levels of phosphodimethylaminoethanol increased to a maximum at 1-2 h and decreased afterwards, whereas concentrations of phosphatidylethanolamine increased continuously throughout the 7 h observation period. This study further found that after i.p. injections of labeled dimethylaminoethanol, the brain content of phosphatidylethanolamine increased through the 7 h period and the levels were 10-40 fold higher than those of phosphodimethylaminoethanol. |
|
Purification Methods |
Dry the amine with anhydrous K2CO3 or KOH, and fractionally distil it. [Beilstein 4 IV 1424.] |
|
Definition |
ChEBI: N,N-dimethylethanolamine is a tertiary amine that is ethanolamine having two N-methyl substituents. It has a role as a curing agent and a radical scavenger. It is a tertiary amine and a member of ethanolamines. |
|
General Description |
A clear colorless liquid with a fishlike odor. Flash point 105°F. Less dense than water. Vapors heavier than air. Toxic oxides of nitrogen produced during combustion. Used to make other chemicals. |
|
Industrial uses |
Dimethylaminoethanol is used as a chemical intermediate for antihistamines and local anesthetics; as a catalyst for curing epoxy resins and polyurethanes; and as a pH control agent for boiler water treatment. However, dimethylaminoethanol in the salt form, (i.e. dimethylaminoethanol acetamidobenzoate) is primarily utilized therapeutically as an antidepressant (HSDB 1988). |
InChI:InChI=1/C4H11NO/c1-5(2)3-4-6/h6H,3-4H2,1-2H3
-
-
Primary and secondary amines are N-methy...
-
A scrubber system was developed specific...
The syntheses of four head group labeled...
-
The kinetics of hydroxyethylation of dim...
The invention provides N-N - dimethyl mo...
A process for preparing a N-substituted ...
Provided herein are a RET inhibitor, a p...
Catalytic hydrogenation of esters is ess...
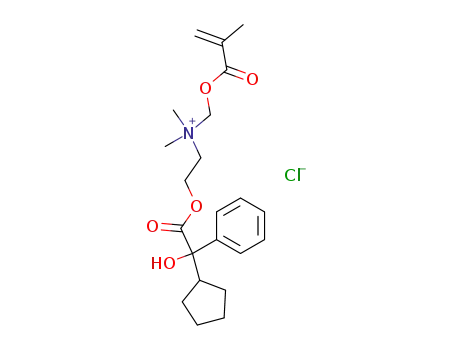
Cyclozil chloromethacryloyloxymethylate

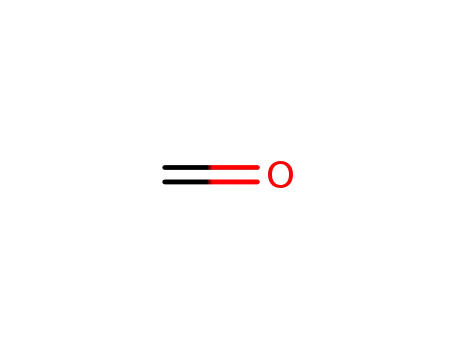
formaldehyd

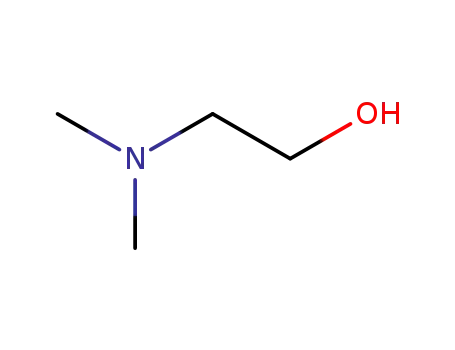
2-(N,N-dimethylamino)ethanol

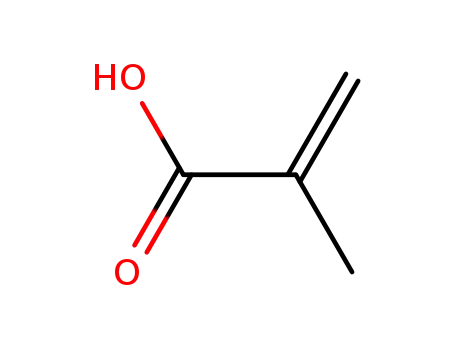
poly(methacrylic acid)

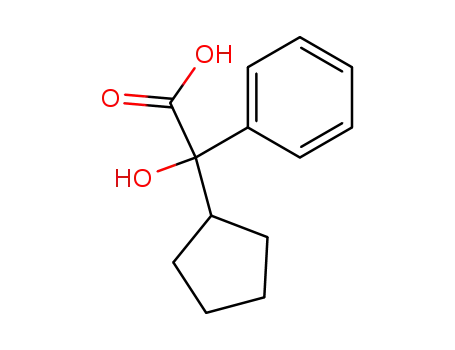
2-cyclopentyl-2-hydroxy-2-phenylacetic acid
| Conditions | Yield |
|---|---|
|
With
potassium chloride;
In
methanol;
at 24.9 ℃;
Rate constant;
Kinetics;
Mechanism;
different pH values from 298-323 K, different ion strength of KCl solution;
|
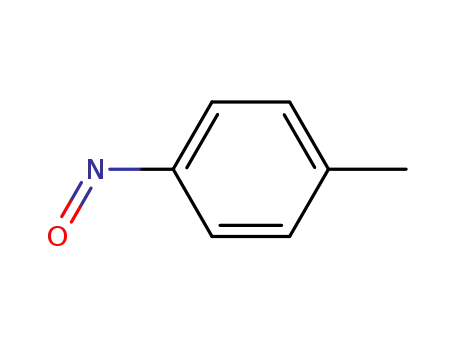
1-methyl-4-nitrosobenzene

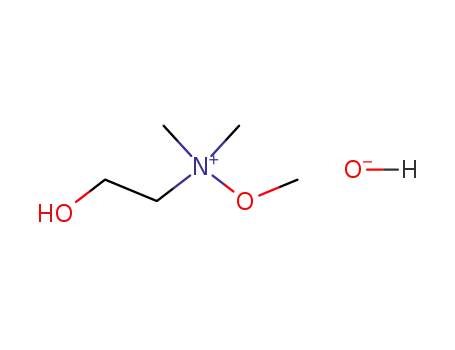
(2-hydroxy-ethyl)-methoxy-dimethyl-ammonium; hydroxide

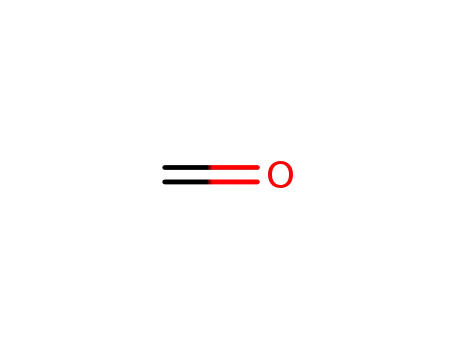
formaldehyd


2-(N,N-dimethylamino)ethanol
| Conditions | Yield |
|---|---|
|
|
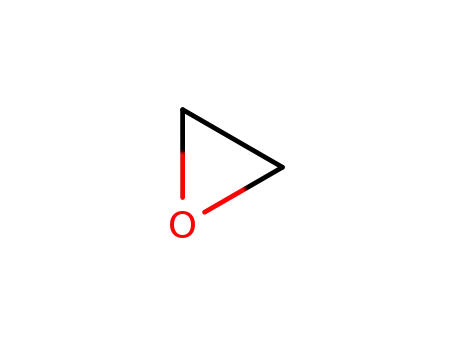
oxirane
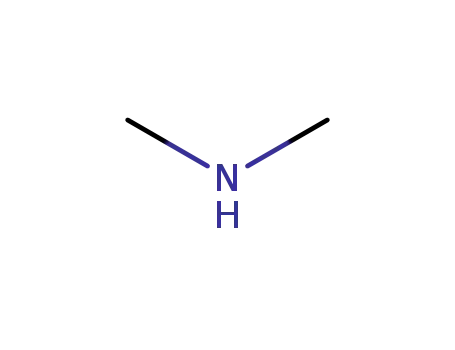
dimethyl amine
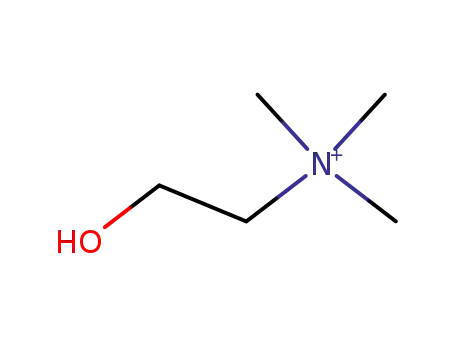
choline
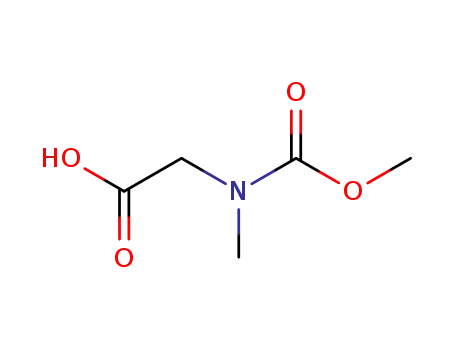
N-methoxycarbonyl-N-methyl-glycine
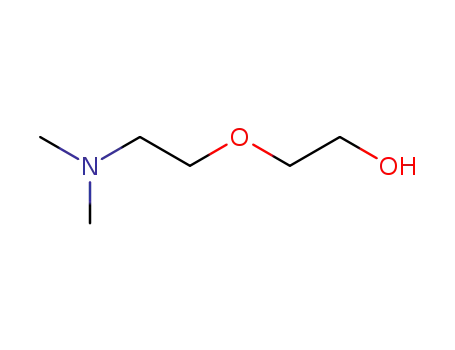
1-O-Dimethylaminoethyl-ethylenglykol
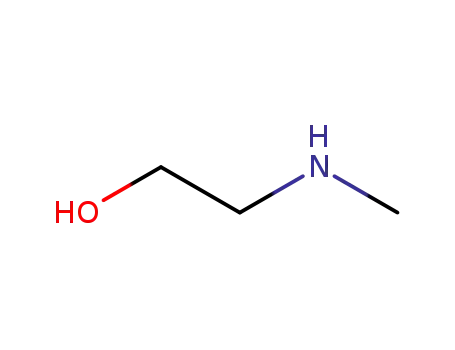
(2-hydroxyethyl)(methyl)amine
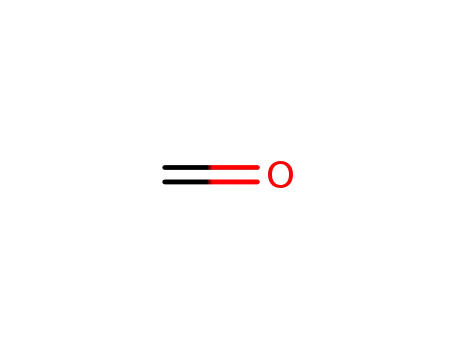
formaldehyd
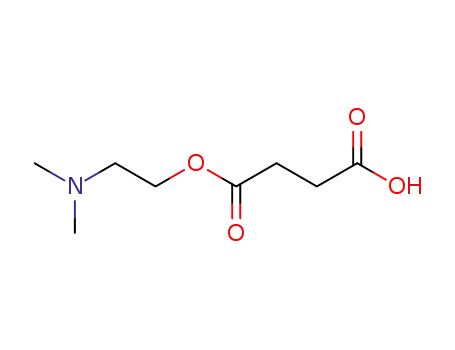
DMAE succinate